- Ship Hull Sheathing
- Sea Service Evaluations
- Sheathing Methods and Cost Analyses
- Sheathing of Offshore Structures
- References
Ship Hull Sheathing
Background
Marine engineers have sought effective and economical means to protect ship hulls and marine structures from the ravages of sea water and marine life for centuries. Wooden hulled ships were sheathed with lead by tramp coastal traders in the time of Alexander the Great. Lead, like copper, resists biofouling growth.
The British Admiralty used sheets of copper nailed to wooden ships starting with the frigate Alarm in 1761 to protect against worms and fouling. Galvanic corrosion of iron fasteners and bolts led to loss of the copper and weakening of the ships. It was not until the 1780s with the introduction of copper fasteners that copper sheathing became popular. With the advent of steel hulls, copper sheathing gave way to copper-based antifouling paints.
The ideal hull material for ships of various sizes and duties has been the subject of intense study, and a variety of hull materials are being used or have been evaluated. Copper-nickel as a hull material was first used as early as 1941 in the construction of Miss Revere, a 13.7 m yacht with a 2mm thick copper-nickel hull. Copper-nickel as a hull material was first used in the mid-1960s by a professor at the University of Alabama after he made an extensive study of a wide variety of possible hull materials for his yacht, the Asperida. He had been seeking material with the best combination of corrosion and biofouling resistance. Alloy C71500, 70-30 copper-nickel, was selected over C70600 90-10 copper-nickel, because the greater strength was an advantage in his particular design.
This led directly to a series of engineering studies by the International Copper Research Association (INCRA - now the International Copper Association), the International Nickel Company and the Copper Development Association Inc. and the construction in 1971 of Copper Mariner (Figure 1), a 20.4 m commercial shrimp trawler with a copper-nickel hull. The primary objective was to determine whether the inherent resistance of alloy C70600 to biofouling and corrosion would generate sufficient fuel and maintenance savings to justify the cost premium for the copper-nickel hull.

Figure 1 - The Shrimp Trawler, Copper Mariner
Copper Mariner has been described elsewhere (2) and will not be elaborated on here. This trawler has a 6 mm thick alloy C70600 hull welded to steel framing. Copper Mariner demonstrated a return on investment in the range of 12.9 to 16.7 percent after taxes for shrimp trawler operations off the coast of Nicaragua. These figures are based on measured fuel and maintenance savings and estimates of the increased earnings potential of this boat due to additional days of availability when compared to the sister steel hulled boats.
The corrosion rate of the hull was less than the 0.00125 mm/yr reported for long-term exposure of C70600 (3) . Interestingly, hull plate thickness measurements after 52 months of service showed no differences in metal loss in the heat affected zone areas of the hull where re-solution of the precipitated iron had taken place compared to the hot-rolled plate with iron precipitated. The majority of the hull had a dark-colored corrosion product film; the heat affected zones were a light golden color. The speed of Copper Mariner is in the 4-10 knot range.
The copper-nickel hull of Copper Mariner is very resistant to fouling (Figure 3). Comparison with the steel hull of the sister ship just prior to its third cleaning after 18 months of service, is striking (Figure 2).

Figure 2 - Marine fouling on the steel hull boat, Jinotega, prior to its third cleaning after 18 months of service

Figure 3 - The Copper Mariner is free of fouling
Copper Mariner was 20 years old this year † . She is still in service, and at last word from the Minister of the National Fishing Industry in Nicaragua, the copper-nickel hull has never required maintenance. Inspection of the hull at drydockings for propeller and shafting maintenance have shown no noteworthy signs of general or pitting corrosion. Four similar boats with 4.8 mm- thick C70600 alloy hulls were built in the early 1970s for service in Sri Lanka.
A number of very successful copper-nickel hull ships have been constructed using a composite material of 90-10 copper-nickel alloy clad on to steel. A second shrimp trawler, Copper Mariner II, was built in 1976 with 8 mm, 25% C70600 alloy clad steel hull plate. This hull demonstrated fabricability and weldability using clad material and welding techniques described by Prager et. al. (4) . More recently, the Italian Ministry of the Interior purchased several fire boats for the port at Naples and other locations. These boats have hulls made from 2 mm of 90-10 copper-nickel clad on 6 mm of steel with a length of 22.56 m and a displacement of 72 tonnes. These copper-nickel hulls assure maximum availability of the fire boats by eliminating the need for hull maintenance of any kind.
Two clad-metal-hull pilot boats were constructed in 1987 for the Board of Navigation in Finland. These two boats were planned as a comparison between austenitic stainless steel and 90-10 copper-nickel for use in the Baltic Sea where ice is a major problem. Both boats are reported to be performing well.
Two boats of note have been constructed in Japan. Although we have no details, we were informed of a yacht, the Akitsushima, built by the Furukama Electric Co., Ltd., with a 4-mm-thick copper-nickel sheath welded to the steel hull. In 1989, this boat was reported to have crossed the Pacific Ocean to Victoria, British Columbia, Canada. The NKK Corporation has reported the construction of an experimental ship, Cupro, with a copper-nickel clad steel hull. These Japanese ships are of great interest and we would be most interested in knowing about their performance and construction details. A summary of vessels constructed to date with copper-nickel hulls is presented in Table 1. The concept has attracted considerable attention for smaller commercial craft, but a ship of length greater than 25 m has not been built.
† This article was written in 1991
| Vessel | Hull Length (m) | Launched | Built | Thickness (mm) | Operating |
|---|---|---|---|---|---|
| Miss Revere | 13.7 | 1941 | USA | 2 | -- |
| Asperida II | 16 | 1968 | Holland | 4 | USA |
| Ilona | 16 | 1968 | Holland | 4 | Curacao |
| Copper Marina | 22 | 1971 | Mexico | 6 | Nicaragua |
| Pink Lotus | 17 | 1975 | Mexico | 4 | Sri Lanka |
| Pink Jasmine | 17 | 1975 | Mexico | 4 | Sri Lanka |
| Pink Rose | 17 | 1975 | Mexico | 4 | Sri Lanka |
| Pink Orchard | 17 | 1975 | Mexico | 4 | Sri Lanka |
| Copper Marina II | 25 | 1977 | Mexico | 6+2* | Nicaragua |
| Sieglinde Marie | 21 | 1978 | UK | 6 | UK/Caribbean |
| Pretty Penny | 10 | 1979 | UK | 3 | UK |
| Sabatino Bocchetto | 21.5 | 1984 | Italy | 6+2* | Italy |
| Romano Rosati | 21.5 | 1984 | Italy | 6+2* | Italy |
| Aldo Filippini | 21.5 | 1987 | Italy | 6+2* | Italy |
| Fire Boat VF544 | 21.5 | 1897 | Italy | 6+2* | Italy |
| Pilot Boat | 14.4 | 1988 | Finland | 7+2.5* | Baltic |
| Akitswushima | NA | 1989 | Japan | steel+4** | Japan |
| Cupro | NA | 1991 | NKK Japan | 4.5+1.5* | Asano Dockyard |
| * Copper-nickel clad steel plate ** Copper-nickel sheathed steel hull NA = not available |
|||||
Cladding and Sheathing
For larger ships, solid copper-nickel hulls are obviously impractical. Practical means of facing the steel with copper-nickel include both cladding and sheathing. By cladding we mean a composite material having a metallurgical bond between the steel and the copper-nickel alloy. Both hot-roll bonding and explosive forming have been used to produce this metallurgically sound interface. Sheathing refers to the attachment of relatively thin copper-nickel sheet to the hull by welding or the use of adhesives. Sheathing is therefore applicable to retro-fitting existing ships as well as to new construction. Sheathing, with or without an insulating layer between the copper-nickel and the steel, is also applicable to off-shore oil and gas structures as well as a wide variety of bridge, dock structures and power or chemical plant sea water intake systems for corrosion protection and prevention of biofouling. Sheathing with copper-nickel is the main thrust of this paper because of its apparent relatively attractive economics and its flexibility and applicability to a wide variety of marine structures in addition to ship hulls.
As noted at the outset in this paper, sheathing is an old concept. Copper-nickel as a ship hull material, as verified in the experiences of the many smaller craft built to date, does, as has been claimed, provide corrosion protection and eliminate fouling of the hull, both of which contribute to reduced requirements. But these concepts have not yet been combined on a large ocean-going vessel to effect the advantages observed when the concept is applied to smaller craft. This may be because a variety of technical and economic issues remain unresolved in the minds of members of the ship building community.
We will examine two issues relating to corrosion behavior of copper-nickel ship hulls which are frequently raised, namely the erosion-corrosion capability of the copper-nickel alloys and the likely consequences of the galvanic corrosion of the steel hull in the event of damage to the sheath. The more complex issue of the life-cycle economics of sheathing will be analyzed for a particular ship. Results of a study of sheathing methods and costs for large ships used in this analysis is reviewed. Shipbuilding Co. in the United States will be reviewed.
Sea Service Evaluations
Erosion-corrosion of Copper-Nickel and the Question of Velocity Limitation
Copper Mariner and many of the other ships listed in Table 1 have maximum velocities in the range of 10-12 knots (5-6 m/s). Certainly, alloy C70600 has demonstrated excellent corrosion resistance both to pitting and to erosion-corrosion in seawater at these velocities on the hulls of these ships. It remained to be demonstrated that alloy C70600 had adequate tolerance at velocities in the range of 25 knots (12.5 m/s) required of large tankers or freighters. Although the maximum velocity recommended for 100 mm and larger pipe is 3.5 m/s, there was indication that the corrosion rate in pipes of very large diameter, or on the essentially flat plate geometry of a ship's hull, might be quite acceptable at very much higher velocities.
Experiments on large ships were performed to try to directly assess this velocity effect (5) . First, a complete rudder was sheathed and installed on the Great Land, a 24-knot roll-on/roll-off vessel. The rudder area is a region of complex and turbulent flow. The tests show that alloy C70600 and the Cu-Ni-Cr alloy C72200 are extremely durable even in the conditions of Cook Inlet off the coast of Alaska, where ice and abrasive silt in the water are encountered. The rudder with 74 m2 of copper-nickel alloy sheathing is shown in Figure 4.
 Figure 4 - Rudder of the Great Land sheathed in copper-nickel
Figure 4 - Rudder of the Great Land sheathed in copper-nickelCorrosion rates were determined by measuring sheathing thickness at intervals of several months up to 14 months of service. Thickness losses were very much a function of position on the rudder as would be expected, but in no position did the thickness reduction exceed 0.1 mm over the 14-month test period. More importantly, the mean loss rate was observed to decrease with time as shown in Figure 5. Mean corrosion rates of less than 0.025 mm/yr at the 14-month point were experienced for both alloys. This means that a stable protective film was being formed on the rudder sheathing preventing the potentially disastrous high corrosion rates thought possible by some people in this high velocity, turbulent regime.
mils/year
Time (Months after delivery)
Figure 5 - Mean corrosion loss rate vs. time for copper-nickel sheath of the Great LandSince galvanic interaction between the hull of the Great Land and its sheathed rudder may have affected the corrosion results, tests were next performed with copper-nickel panels attached to, but electrically insulated from, the hull and the rudder. Panels with exposed faces 250 x 125 mm and insulated with Teflon and epoxy (Figure 6) were assembled and fastened to the ship at four locations 4.6 m below the water line. Measurements indicated that some of the panels developed partial grounding to the hull, meaning that in these cases the absolute value of the corrosion rate is probably lower than it would have been had the insulation remained undamaged. The results obtained by thickness measurements showed no significant difference in the corrosion rate of the panels at the various hull locations. The rudder panels exhibited higher erosion-corrosion rates than those on the hull (less than a factor of 2) and the rates were higher on the insulated rudder panels than on the corresponding sheathed-rudder test locations. Cathodic protection of the sheathed rudder by the hull had suppressed the corrosion rate by about a factor of three as shown here:
| Loss Rate (mm/yr) | ||
|---|---|---|
| Alloy | Sheathed Rubber | Insulated Panels On Rudder |
| C70600 | 0.02 | 0.08 |
| C72200 | 0.04 | 0.10 |
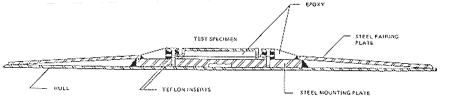
Figure 6 - Schematic of method for insulating and attaching copper-nickel panels to the hull of the Great Land.
The C72200 Cr-containing alloy consistently showed slightly higher corrosion rates. This was not as expected and it is not certain that the observed difference is significant. The corrosion rate data for the insulated panels showed a definite decreasing tendency with time, again indicating that a stable protective film was forming over the 14-month period of the tests. The rates were tending to values of 0.02 to 0.1 mm/yr (and presumably lower if measurements after longer exposure times had been possible), which would be quite acceptable in this application.
Based on these observations, it is believed that 12.5 m/s (24 knots) is below the breakaway velocity for copper-nickel in ship hull situations. It is noted that the breakaway velocity determined by jet impingement testing in the laboratory for alloy C70600 was approximately 5 m/s (10 knots). It is possible that the differences in laboratory and ship hull results can be attributed to factors such as:
- Intermittent flow on a ship hull versus continuous exposure in the laboratory;
- Differences in hydrodynamic flow conditions in the two situations; and
- Differences in dissolved oxygen and temperature of the sea water in the laboratory compared to the actual hull exposure conditions experienced.
No matter what factor or factors predominate, it is felt that the panel tests are more predictive of actual hull performance than laboratory tests since the hull panels duplicate the actual ship service conditions.
Sheathing Attachment Methods
In the early 1980s, another hull panel experiment was performed to test attachment methods and, again, to evaluate the performance of copper-nickel panels on a ship in service (6) . A group of panels were attached to the hull of the Arco Texas, a crude-oil tanker, during construction of this ship at Newport News Shipbuilding, Inc. This ship was designed to be, and in fact is, the largest crude carrier capable of passage through the Panama Canal. Although not initially a part of the exposure plan, the severe abrasion and impact loading attending passage through the Canal provided a realistic, perhaps "worst case" test situation.
Twelve copper-nickel panels, each 0.9 x 3 m and 3 mm thick were divided into four sets of three for emplacement on the hull near the light load line as shown in Figure 7. The panels were thus subjected to fully submerged, alternately wet-dry and splash zone conditions. One group was attached by peripheral welding using ENiCu-7 SMAW electrodes reinforced by bonding to the hull with Boskik M-890 epoxy. A second group was attached by peripheral welding with no interior reinforcement. A third group used peripheral welding reinforced with 3M XB-5354 elastomeric adhesive. The last group was attached with the peripheral welds supported by an array of equally-spaced slot welds across the face of each panel.
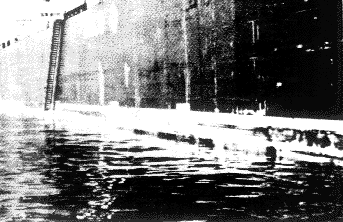 Figure 7 - Location of copper-nickel panels on the Arco Texas
Figure 7 - Location of copper-nickel panels on the Arco TexasAfter two years in service in a wide spectrum of sea service conditions including warm Gulf waters, a number of trips throughout the Panama Canal, and West Coast waters from Southern California to Alaska, all panels were intact. All panel and welds were tightly adhered to the hull and leak free. There was evidence from magnaguage readings that the epoxy and elastomeric adhesives had debonded to some extent during service. These forms of reinforcement, therefore, received no further consideration. Several panels had rather severe scratches and gouges from contact with sides of the Canal, docks or tugs. Thickness measurements before and after service indicated the corrosion loss to be less than 0.025 mm, corresponding to a maximum corrosion rate of less than 0.0127 mm per year. Apparently, the corrosion life of the panels is far in excess of the service life of the vessel.
No evidence of biofouling was apparent on the copper-nickel panels after the two years. The fact that the copper-nickel remained antifouling implies it was also freely corroding during this period, albeit at a very slow rate. Therefore, the panels could not have been provided significant cathodic protection by the surrounding steel hull and they could not have contributed to corrosion of the steel hull, even though the hull paint system immediately around the panels, repaired after installation, had failed to some extent. The hull was host to fouling and after two years had a great deal of algal "grass" several inches-long attached.
Sheathing/Cladding Damage and Galvanic Corrosion of the Hull
The question of rapid and potentially disastrous corrosion of the steel hull plate in the event of damage to the copper-nickel sheathing or cladding has been raised since cladding and sheathing concepts were first introduced. The mean, free corrosion potential of steel is anodic to that of copper-nickel by about 300 mV. Drodten and Pircher at Thyssen Steel AG in Germany carried out tests to evaluate this problem by mounting copper-nickel clad on steel test pieces in the immersion zone of a test rig on Helgoland and on the hull of a sea-going tug (7) .
The test pieces were mounted so as to be electrically insulated from the steel test rig or tug boat hull. Holes and slots were machined through the copper-nickel to expose the steel to provide a variety of defect sizes and a range of cathode-to-anode surface area ratios. Defect-free specimens were exposed as well. These were found to be rust-free and free of fouling after exposure. The tug boat was in service at the mouth of the river Ems and, for a period in the cold winter of 1986-87, was used also for ice breaking. The test plates were assessed in a dry dock at intervals of a few months. Individual test pieces were removed for laboratory examination and new ones attached. Some of the test pieces were exposed over the entire test period of 16 months.
Test pieces with larger defects, such as the 5 x 100 mm2 slits, were found after three months' exposure to have profuse rust and fouling. Because of the cathodic polarization of the copper-nickel caused by exposure of the steel at the slit, the fouling resistance of the copper-nickel had, as expected, been lost. Cross-sectioning of the corroded slit defect showed the steel to be undercut by corrosion to about twice the width of the slit in the copper-nickel cladding.
After 4.5 months of service, the test pieces were found not to have the heavy rust and fouling. Again, test pieces were removed and sectioned through the corroded defect in the copper-nickel. Clear differences in the depth of corrosion and in the undercutting, depending on the size of the defect, were found. The 5-mm and 10-mm diameter defects displayed corrosion depth of about 6-7 mm. At the 50-mm diameter defect, on the other hand, the corrosion depth was only 1.5-2 mm.
After a further period of eight months, the tug boat tests were concluded and all test pieces examined in the laboratory. Corrosion depth and undercutting were measured. On the copper-nickel itself, no thickness reduction caused by corrosion, erosion or abrasion was evident after 18 months of ship operation. The original surfaces with the shallow grooves from specimen preparation were practically unchanged.
At Helgoland the test pieces were subjected to one-year of undisturbed exposure in the immersion zone with no interim examinations. Upon removal, light barnacle fouling was found on the surface. The surface at the larger defects exhibited dense marine growth and was covered by dark corrosion products.
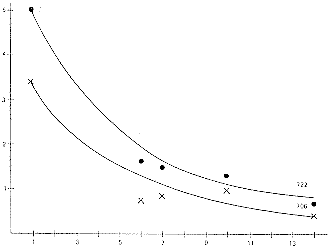
The measured corrosion rates and anodic current density values calculated from Faraday's Law are plotted in Figure 8 against the area ratios. The influence of area ratio Sc/Sa is unmistakable. The corrosion rate increases up to a limiting value at about Sc/Sa=140. Corrosion rates are higher for the tug boat test pieces in the winter, apparently due to the higher oxygen level of the colder sea water. The water in winter at the mouth of the Ems River contains 11-13 ppm oxygen at about 0°C and only about 5 ppm when water temperatures are 18-20°C in the summer. The corrosion rates are in very nearly the same ratio as the seasonal oxygen level, suggesting that the corrosion reaction is controlled by the rate of the cathodic reduction of oxygen reaction.
i a(mAcm -2)
w(mma -1)
Sc/Sa
Figure 8 - Anodic current densities vs. cathode-to-anode surface area ratios for clad copper-nickel/steel specimens with machined defects to expose the steelThe authors conclude that the probability of the occurrence of through-the-hull damage is very small and only likely in the case of a very small defect.
The corresponding situation involving defects in a sheathed hull is somewhat different, as indicated in an experiment on the corrosion of steel in the vicinity of a defect in the sheathing of a steel piling (28) . A small defect in the copper-nickel sheathing will allow a thin layer of water to enter the arrow space between the sheathing and the steel hull bounded by the peripheral sheathing welds to the steel. The Sc/Sa area ratio will be much smaller than the apparent geometric ratio. Corrosion of the steel will proceed rapidly for a very short period until the oxygen is consumed because the entrapped film of water has very poor contact with the outside sea water. It would appear that this situation is less threatening to the integrity of the hull and will allow more than ample time to make a repair to the sheathing.
Hull Roughness
A most important reason for using copper-nickel on the hull of a ship, either by cladding or sheathing, is to achieve fuel economy by virtue of the reduced hull roughness achieved with copper-nickel. The hull roughness is directly related to the shaft horsepower required to overcome simple hull friction; the rest of the power is expended in wave making.
It has been estimated that a shipowner pays a penalty equivalent to a one percent increase in power requirement for an increase of 10µm in hull roughness (in the range 0-230 µm) and 0.5 percent for every 10µm beyond that (8,9) . Based on a British study of 400 ships, the average roughness of a new hull is 125µm, the best attainable roughness is 75µm, and the average yearly increase in roughness ranges from 50 to 70µm. A conventional hull, therefore, begins to lose efficiency from the day it is launched, while a copper-nickel hull, if there is any change, contributes to increased efficiency with time.
The roughness data enable calculation of probable ship performance if the entire wetted surface had been sheathed with copper-nickel. At the end of the test period, the mean roughness of the painted steel hull was 250µm. The corresponding roughness of the copper-nickel was only 53µm. The difference, 197µm, represents the improvement in operational efficiency attainable if the entire hull is sheathed. Assuming the power penalty estimate cited above is valid, full-wetted-surface sheathing with copper-nickel panels would result in a 19.7 percent improvement in efficiency. An independent study conducted by the Webb Institute supports these findings (10) .
Roughness measurements of the 12 panels of the Arco Texas experiment showed that the copper-nickel is considerably smoother at the outset than a painted steel hull, and of considerable interest, the copper-nickel appeared to become slightly smoother after the sea trials. Painted steel, of course, becomes progressively more rough in service. Hull roughness data measured at the end of the Arco Texas exposure are shown in Figure 9.
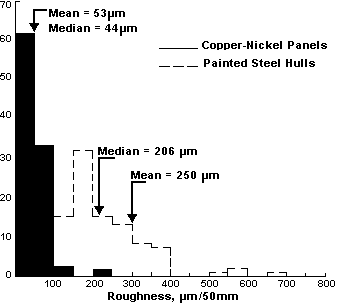 Figure 9 - Final roughness data at end of in-service testing of copper nickel C70600 panels on the Arco Texas
Figure 9 - Final roughness data at end of in-service testing of copper nickel C70600 panels on the Arco TexasCopper-nickel sheet for sheathing, because it is cold rolled, is somewhat smoother than the hot- rolled clad material. Cold-rolled material typically has a mean roughness of about 40µm; whereas the hot-rolled clad material typically has a mean roughness of about 60-55µm. Determination of the roughness of the copper-nickel sheathing on the rudder of the Great Land showed the roughness to be consistently lower than 20µm. For comparison, the roughness of the painted steel hull of the ship measured on dry docking after 14 months averaged approximately 210µm.
Sheathing Methods and Cost Analyses
State-of-the-Art Baseline Method
To effectively analyze the potential operational and maintenance savings for a large ship associated with having a corrosion and fouling resistant copper-nickel hull, it is necessary to have a sound estimate of the cost of sheathing. This analysis was undertaken by the Newport News Shipbuilding Company for the Copper Development Association under a contract from the U.S. Maritime Administration (MARAD) (11) .
A ship was first selected for analysis. To suit the purpose of this study, it was determined that a vessel that could be related to both ends of the spectrum of high-block, slow speed ships to low-block, high speed ships would be a desirable choice. For their analysis, Newport News Shipbuilding selected the fleet oiler T-A0 187 class, T-A0 191 series ship as the baseline ship for the study. The block coefficient, wetted surface, and basic ship configuration lend themselves well to comparison with various other vessel types to determine the approximate cost of sheathing of other ships. The dimensions and pertinent characteristics of the fleet oiler are shown in Figure 10. The maximum loadline of 38 ft. (11.6 m) above baseline was taken as the line of demarcation for sheathing; i.e., the entire hull below this line would be sheathed including the rudder and sea chests. The sea chests however were not included in the calculations. This gives 83,042 ft 2 (7,715 m 2) of hull to be sheathed.
| Length Overall | 677' - 6" |
| Length between perpendiculars | 650' - 0" |
| Beam, maximum | 97' - 6" |
| Draft to design waterline | 34' - 6" |
| Cb | 0.643 |
| Displacement | 40140 Long Tons |
 Figure 10 - Fleet oiler T-AO 187 class T-AO 191 series profile
Figure 10 - Fleet oiler T-AO 187 class T-AO 191 series profileThe sheathing alloy was taken to be copper alloy C70600. The sheet-size selection had to consider the ability to handle and position the sizes commercially available. For the initial analysis, a sheathing sheet size of 48 in x 144 in x 0.1 in (122 cm x 366 cm x 2.5 mm) was chosen. Each sheet would weigh 223 lbs (101 kg). The total welding required was approximately 55,000 linear feet (16,764m). A total of 1,731 full size sheets weighing 424,874 lbs (193 tonnes) would be required. Allowing for cutting to fit around the bow and stern, it was estimated that a total of 2,000 sheets would be necessary.
Based on the weld attachment studies described above, manual SMAW using AWS A5.11 class ENiCu welding electrodes were initially considered. The horizontal plate spacings would be 3/8 in (9.5 mm) and 5/8 in (15.9 mm) on the vertical joints.
An array of 1 in x 3/8 in (25 mm x 9.5 mm) slot welds would be used to strengthen the plate attachment to prevent panting of the sheathing. Section 6 of the American Bureau of Shipping Rules for Building and Classing Vessels was used to determine the hull girder bending stresses. These analyses were conducted at the 1/4, mid and 3/4 sections of the hull. The calculated bending moments, combined with section modulus at midship, provided the magnitude of the stresses that act on the vessel about the neutral axis. The frequency of slot welds is dependent on both the calculated stress and the strength of the copper-nickel to steel weld. These calculations indicated that the frequency of slots could be reduced the closer the sheets were to the neutral axis. The results showed that on two thirds of the hull surface, the slot spacing should be 12 in (300 mm), and on the remaining one third, the spacing could be increased to 16 in (406 mm). This results in a total of 26,468 slot welds on the ship, and, at least to a first approximation, minimizes the number of slot welds required.
Consulting representatives of various construction departments at Newport News Shipbuilding established the following construction sequence for sheathing:
- Dock vessel
- Blast and coat - shot/sand blasting and coating with thin coat of rust preventative
- Erect Staging - complete seven-tier staging would be required
- Grind Flush Existing Welds - estimated to be 17,000 linear feet (5,182 m)
- Non-Destructive Test of Existing Welds might be necessary in some cases - cost not included in this estimate
- Install Sheathing - mark off, remove coating, tack weld
- Weld Sheathing - peripheral and slots
- Grind Flush Sheathing Welds - 27,000 linear feet (8230 m)
- Remove Staging
- Fleet Vessel
- Non-destructive Test Sheathing - vacuum test
- Undock Vessel
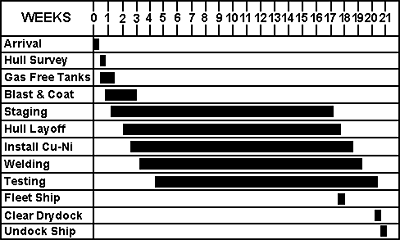
The time and personnel required for each operation was established. As shown in Figure 11, a period of 21 weeks was considered realistic for sheathing. Costs were then estimated for each of the steps above. Information on man-hours to complete a task, length of time in dry dock, unit of time per square foot of area, price for the sheathing material and welding electrode, etc., was required. Results broken down into percentages of total cost are shown in Figure 12. In terms of a unit price, the cost was calculated to be $56.54 per sq.ft. 2 ($608.59/m 2) (in 1986 dollars). If NDT of the existing steel hull welds is required, this could add $0.30/ft 2. ($3.23 per sq.m) of coverage area.
Improved Sheathing Method
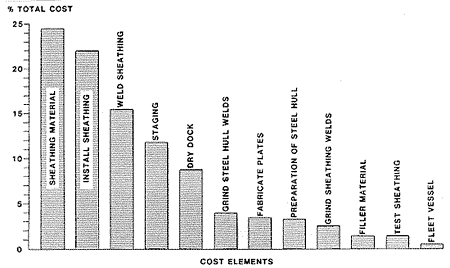
It was recognized that invoking some innovative technology for critical portions of the state-of-the-art approach used above had the potential for substantially lowering the cost. The five major "cost drivers": sheathing material, 25%; install sheathing, 22%; weld sheathing, 15%; staging, 11%; and dry docking, 9%, were analyzed for potential improvements. Three other areas were examined and chosen as secondary areas for improvement, the grinding of existing hull and sheathing welds, and the preparation of the steel hull.
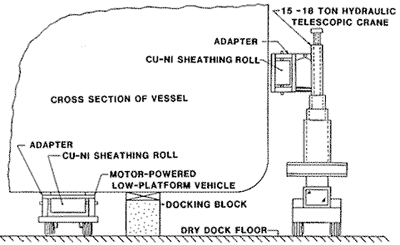
First, the substitution of coiled copper nickel for individual sheets would result in reduced material expense and benefits in lay-off of the hull and placement of material on the vessel. A 24-in-wide coil weighing 7,000 lbs (,3176 kg) would have a length of over 750 ft (229 m) and could be handled by a special rig to support the coil and allow the material to be unwound against the hull during installation (Figure 13). Such a rig should be designed so it could be attached to a typical 15-18 ton hydraulic telescope crane and to the motor-powered, low platform vehicles seen in most shipyards. This equipment would minimize the requirement for the detailed hull layout and facilitate initial placement of the sheathing on the ship. The reduced costs of these items and the material would be partially offset due to the increased linear feet of welding (approximately 2O%) because of the smaller width. Semi-automatic welding machines could be used in conjunction with these rigs to tack-weld the sheathing in place. Another significant area of saving, inherent to the use of coil sheathing material, is that this allows most of the expense for staging to be eliminated. Mobile elevated platforms supporting the coils would also allow workers access to any area of the vessel and allow work to progress without lag time for erection of staging.
On-going research on welding methods indicated the Metal Inert Gas (MIG) Beam welding technique to be a potentially viable method (12). The MIG Beam process is a coil-fed, semiautomatic technique requiring only a 1/8 in (3.2 mm) gap which can be filled in a single pass rather than the three passes required for the wider gaps in the SMAW process. The use of semiautomatic equipment and the single pass reduces the man- hours of labor to complete the task.
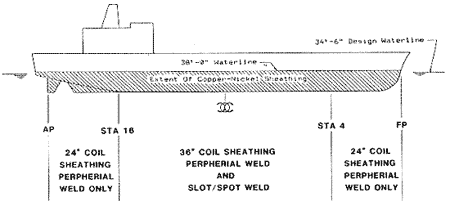
Further investigation into the hull girder bending stress of this vessel indicated areas of reduced stresses where the requirements for attaching sheathing to the vessel would be less stringent. As shown in Figure 14, approximately 30% of the coverage area required peripheral welds only if 24-in (0.61-m) wide sheathing was used. In the mid-body section of the vessel, 36-in (0.91-m) wide material could be used. This region would require a series of slot welds consisting of a 12-in (0.61-m) center-to-center spacing for about 80% of the coverage and 18-in (0.46-m) spacing for the remaining 20%. This configuration reduces the number of slots required from 48,468 to 34,878. The 1 in x 3/8 in (25 mm x 9.5 mm) slots by manual SMAW would again be used.
Another possible area of improvement is to use spot welds. A pulsed MIG spot welding technique developed (12) , has proven effective in all positions. A 3/16-in (4.8-mm) diameter hole with an 82° taper is punched. Three spot welds for each slot weld are required to carry the same load, but the welds could be done semiautomatically. This would eliminate all manual welding on the sheathing and allow for a faster and more effective attachment process requiring a minimum of highly skilled personnel.
Another area where costs can be reduced is in eliminating the grinding of welds. The MIG Beam process does not create large weld beads and would not pose any significant degradation in hull performance. As for the steel hull welds, the copper-nickel sheathing is sufficiently flexible and would essentially conform to the shape of the hull. Only large raised areas need be ground off.
Finally, costs could be saved in hull preparation. The first cost estimate of this study involved sand/shot blasting the entire hull (below the 38 ft. waterline) to white or near-white metal and then applying a thin coating to prevent rust prior to sheathing. Depending on the age of the vessel and the condition of the hull surface at the time of installing the sheathing, only those areas to be welded (butts, seams and slots/spots) need to be cleaned to white metal. Laying off the hull, prior to placement of the copper-nickel sheathing (to indicate the exact locations of the areas to be welded) and then blasting only those areas, would facilitate the process of installation.
Recent developments using the concept of high pressure water as a medium for blasting clean a ship's hull have led to technology that allows specific areas to be cleaned. This system operates by the use of a hand-held, self recovery nozzle that can be limited to a coverage area of only a few inches. Using this or a similar system would allow the areas that are to be welded to be cleaned just prior to the sheathing installation and, therefore, would not require any special treatment to prevent rust.
A revised schedule for sheathing the ship was calculated based on the several improved methods noted above. The total duration for sheathing the vessel is reduced with the improvements to 17 weeks (Figure 15). A new cost analysis incorporating these improvements was performed. Costs for the various steps as a percentage of the total cost are shown in Figure 16. The final cost estimates based on the improved method was established first for a scenario that incorporates the series of 1 in x 3/8 in (25 mm x 9.5 mm) slot welds welded using the manual SMAW technique.
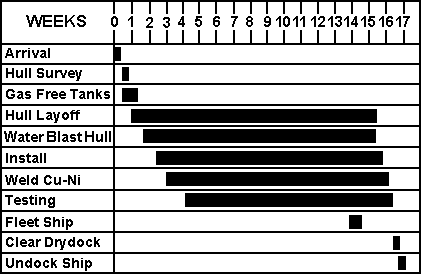 Figure 15 - Improved method construction schedule
Figure 15 - Improved method construction schedule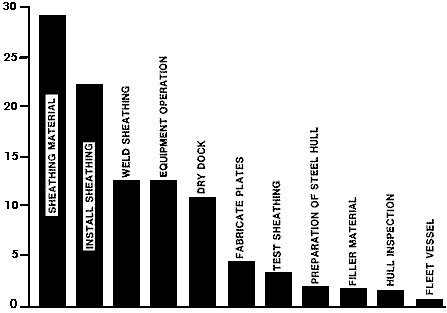 Cost Elements
Cost Elements Figure 16 - Improved method cost estimate
This leads to a unit sheathing cost of $35.28/ft² ($379.61/m²). If the slot welds are replaced by a series of 3/16-in (4.8-mm) diameter spot welds using the semiautomatic pulsed MIG technique, the unit cost is reduced to $33.18/ft² ($357.02/m²). A third scenario worth noting is for sheathing during initial construction of the vessel, where certain costs, including drydocking charged to sheathing installation, are eliminated. In this case, the unit cost of sheathing is calculated to be $25.40/ft² ($273.30/m²). All of these unit costs are in 1986 dollars and are exclusive of NDT of $0.30/ft² ($3.23/m²).
The impact of the improved methods on the individual operations of sheathing is illustrated in Figure 17. Large percentage savings result from the reduced requirements for grinding steel hull welds and sheathing attachment welds. The cost of testing the sheathing in this new configuration is the only operation that is more expensive.
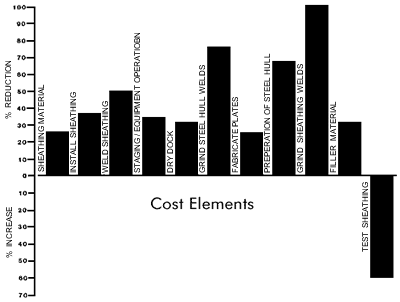
- In order to maintain comparison of the two cost analyses, Staging and Equipment Operation are treated as the same cost element
- Fleeting of the vessel remains the same in both analyses and therefore is not shown on this chart
- Percent difference is based on Baseline Cost Analysis versus Improved Method Costs Analysis cost figures
Life Cycle Performance Economics
The objective of these studies was to determine the effect of the following three hull- coating systems on the total economics of a sea-going vessel.
- Conventional copper-bearing anti-fouling paint with primer and CP system
- Organo-tin copolymer antifoulant (OCA) system
- Copper-nickel sheathing
Two scenarios for sheathing were considered:
- Having the benefit of sheathing for the full service life of the ship; i.e., sheathing during construction
- Sheathing as a retrofit after 10 years of service
The analysis presented here used the costs for the sheathing described above to update an earlier economic study performed in 1983 (10) . In this study by the Webb Institute, a different ship, a C-4 cargo vessel was considered rather than the T-AO tanker. The C-4 cargo ship is obsolete, but there exists a large body of operating data for it. Analysis of the operation economics for this ship was quite detailed and considered a comparison of a number of factors affecting operating costs and revenues as influenced by the choice of hull protection method.
The cost of sheathing was adjusted for the smaller ship taking into account the reduced installation time due to the smaller wetted area, 46,370 ft² (4,313 m²). The 1983 dollars were inflated to 1986 buying power and the cost of fuel oil was taken as $18/barrel.
In the analysis of sheathing during new construction, life-cycle economics were based on a comparison of such factors as: engine cost (specific engine-propeller combinations were chosen on the basis of expected coating performance), the cost of the coating methods themselves, fuel consumption, normal drydock costs during the 20-year service life, and salvage revenue. Cargo revenue was held constant for all coating systems, a consequence of sizing the engine-propeller combination for each coating method to give the same speed in all cases. The coating's effectiveness is reflected in the size of the engine (a capital cost factor) and the resulting fuel consumption rate.
In the case of the retrofit installation, the life-cycle economic comparison among the three hull protection methods was based on the cost of the coating method, the cost of the extra surface preparation needed to apply the coating at time of retrofit, time out of service, fuel consumption (which would differ for the three methods after installation), drydocking costs during installation, normal drydocking costs during the remaining service years after coating, the cost of coating maintenance, and the added salvage value for copper-nickel at the end of the ship's service life. In retrofitting, the engine size does not change, but fuel consumption would be affected by the coating performance characteristics; i.e., speed. Cargo revenue is, therefore, a function of coating type and is included in the analysis for retrofit coating/sheathing installation.
The results of the discounted cash flow economic analyses are summarized in Table 2 and Table 3. In the installation during new construction case, $1,814,000 is saved as a result of the smaller engine required for the copper-nickel sheathed ship. This partially offsets the substantially higher cost for application of the copper-nickel (even for the lowest cost scenario). But the very large savings in fuel over the 20-year service life results in attractive total savings.
| All costs in thousands of 1986 dollars | ||||||
|---|---|---|---|---|---|---|
| Case | Engine Cost | Coating Cost | Fuel Cost | Normal D.D. Cost | Salvage Value | Total Savings |
| Base (Conv.) |
9,200 | 102 | 20,263 | 715 | -- | -- |
| OCA | 8,237 | 163 | 17,438 | 961 | -- | 3,481 |
| Cu-Ni @$56.54 |
7,359 | 2,622 | 14,841 | 440 | 19 | 5,037 |
| Cu-Ni @$35.28 |
7,359 | 1,636 | 14,841 | 440 | 19 | 6,014 |
| Cu-Ni @$25.40 |
7,359 | 1.178 | 14.941 | 440 | 19 | 6,481 |
| All costs in thousands of 1986 dollars except extra cargo | ||||
|---|---|---|---|---|
| Case | Base (Conv.) |
OCA | Cu-Ni @$56.54 |
Cu-Ni $35.28 |
| Retrofit Coating Cost | 72 | 133 | 2,157 | 1,603 |
| Extra Sufrace Prep. | 115 | 115 | 115 | 115 |
| Time O/O svc. for coating | 42 | 42 | 1,214 | 983 |
| Fuel cost | 20,242 | 20,212 | 18,394 | 18,445 |
| Coating drydock cost | 21 | 21 | 607 | 482 |
| Normal drydock costs | 165 | 165 | 83 | 83 |
| Re-coat at drydock > 10 yr. | 327 | 510 | 220 | 220 |
| Extra cargo rev. | 129,020 | 130,270 | 131,570 | 131,570 |
| Total Savings | (60) | 976 | 343 | 1,562 |
| *CU-NI total includes $19,000 salvage bonus | ||||
Normal drydock costs arise because the American Bureau of Shipping requires a biannual tailshaft inspection, which requires drydocking. The copper-nickel sheathing should only require a hydroblast, if that, at these drydockings. But, in the interest of being conservative, a small material and application cost equal to two percent of the cost of the sheathing the entire ship was added to allow for the possibility of surface damage. The repair costs for the organo-tin and conventional systems are higher.
Thus the application of copper-nickel hull sheathing at the time of new construction will result in a net savings of from $5,037,000 to $6,481,000, depending on the cost of initial installation. For sheathing as a mid-life retrofit after 10 years of service, the savings generated by sheathing and the other coating systems are of course not as large.
The "base" case considered here was taken as a complete cleaning of the steel hull to "white metal" followed by application of a conventional corrosion-resistant and antifouling paint system costing $72,000. The sheathing costs are calculated again, using the unit costs described above. Out-of-service time for the retrofit for this vessel is included as a cost at $14,000/day. Drydocking costs have been broken out for those costs associated with the coating process itself and also those normal drydocking costs which the various coating systems will provoke in the last 10 years of service. The copper-nickel sheathing will require only half the time in drydock required for either the conventional paint or the OCA coating, resulting in an $83,000 lifetime savings in favor of the copper-nickel.
Likewise the labor required to maintain the coatings ("Recoating at D.D." - Table 3) will be considered less costly for the copper-nickel sheathing. Fuel savings in this retrofit case accrue only over the last 10 years of service and are, therefore, not as large as in the new construction case. But substantial extra cargo revenues will result from the smoother hull for the given engine-propeller combination due to higher-speed steaming and fewer days in drydock for maintenance. In the last 10 years of operation, the copper-nickel adds $210,000 to the vessel's revenue compared with a conventional coating system.
The net economic result of the copper-nickel application as a retrofit, is listed in the right-most column of Table 3. Copper-nickel hull sheathing will save the vessel owner between $343,000 and 1,562.00 compared with conventional paint. An OCA system will result in a $976,000 saving. Reapplying conventional antifouling paint in an "as-new" condition will actually result in a loss of $60,000 since the cost is not fully offset by improved economy and greater cargo throughout. Therefore, even in the case of a 10th-year retrofit, it appears that copper-nickel sheathing can provide higher rates of savings than either conventional or OCA systems, provided that the technological improvements discussed earlier are exploited.
Ship Hull Sheathing - Conclusions
This section has attempted to review the history and experience with copper-nickel ship hulls and to discuss the technology as it pertains to the sheathing of large ships. Experiments with sheathing of the rudder and placement of copper-nickel panels on the hulls of freighters and tankers have provided basis for dismissing concerns over the velocity tolerance of alloy C70600. Galvanic corrosion of the steel hull has been shown to take place at a manageable rate with copper-nickel clad steel with defects exposing the steel. It is suggested that sheathed hulls will be even less susceptible to corrosion of the steel hull in the event of rupture of the sheath. Sheathing employing existing methods as well as easily adopted advanced approaches have been described with the associated costs per unit area. Finally an economic comparison for three methods of hull protection are compared in terms of initial capital cost and savings in maintenance and fuel over the 20-year life of the ship or over a 10-year life in the case of a retrofit sheathing in mid-life of the ship. In both cases, copper-nickel sheathing is shown to result in considerable life cycle cost savings.
We hope this review prompts a study of the potential for copper-nickel sheathing of large ships by world shipbuilders and leads to a major demonstration project to advance the copper-nickel ship hull sheathing concept.
Sheathing of Offshore Structures
Introduction
Whereas sheathing of ship hulls for corrosion and biofouling resistance is a concept awaiting a definitive demonstration and test, sheathing of a variety of offshore structures is a proven application for copper-nickel alloys and the nickel-copper alloy Monel (15) alloy 400. This is because offshore oil and gas drilling and production platforms must maintain their structural integrity and resist the effects of not only aggressive sea water but weather and wave action as well.
With the stabilization of oil prices, the commercial climate for field development has improved. Over 700 offshore fields are reportedly slated for development in the 1990-1995 period. Some 188 of these fields involving 275 platforms are in Asia (13) .
A number of summary papers and reviews of the sheathing of offshore structures have been published (14-19). This treatment summarizes the salient points with regard to the benefits of sheathing of large offshore structures, vis-a-vis reduced structural cost due to elimination of fouling and corrosion allowance due to splash zone corrosion, and gives some insight into the economic benefits.
Both the fouling resistance and corrosion protection aspects of copper-nickel sheathing are critical to protecting offshore structures. The attachment and growth of marine organisms can add considerable weight to a structure; but more importantly, the increase in side loads on the structure due to currents, wind and waves is a major design consideration. Marine growth as large as 0.7 to 1.2 m has been seen. Excessive marine growths extend frequently to about 1 m above mean sea level to about 10 meters below. Lesser amounts of growth extend to lower levels. Very large amounts of extra steel structure must be provided to resist the resulting forces. Regular and expensive removal of marine growth by divers using high pressure water is also required. As will be discussed below, sheathing can reduce these costs dramatically and provide overall savings to the platform owner.
Corrosion and Protection of Steel Structures in Sea Water
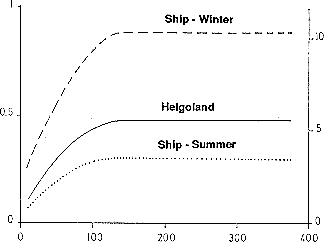
The intensity of corrosion of an unprotected steel structure in sea water varies markedly with position as shown in Figure 18. The spray and splash zone above the mean high tide level is the most severely attacked region due to continuous contact with highly aerated sea water and the erosive effect of spray and tidal action. Corrosion rates as high as 0.9 mm/y at Cook Inlet, Alaska, and 1.4 mm/y in the Gulf of Mexico have been reported. Cathodic protection in this area is ineffective because of lack of contact with the sea water and, thus, no current flow for much of the time. Corrosion rates are often very high at a position just below mean tide in a region that is very anodic relative to the tidal zone due to powerful differential aeration cells.
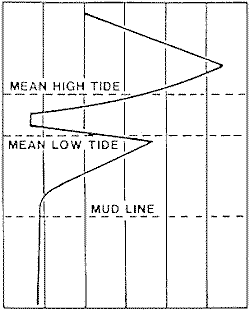 Relative Loss in Metal Thickness
Relative Loss in Metal Thickness Figure 18 - Profile of the thickess loss resulting from corrosion of an unprotected steel structure in seawater
Protection of a steel structure can be effected by various means; each corrosion zone must be separately considered. Three generally accepted methods are cathodic protection, painting or coating, and sheathing. Sheathing has proved to be a very successful approach when applied in the region through the splash/spray zone to a short distance below the tidal zone. Monel alloy 400 was applied to this task as early as 1949 on an offshore platform in the Gulf of Mexico off the Louisiana coast (20,21) . The LaQue Center at Wrightsville Beach, North Carolina, USA, conducted extensive trials of sheathing using the steel piling supporting the sea water corrosion test wharfs at the laboratory as test specimens. Sheathing or protective materials tested included Monel, AISI 304 stainless steel, 70-30 copper-nickel, and nickel-clad and Monel- clad steel. All of these were reported to be performing very well after 36 years of exposure (18) . A large number of proprietary coatings, including galvanizing and sprayed zinc and aluminum, were also tested; all proved to have finite effective lifetimes extending up to 13 years (16) .
The 90-10 copper-nickel alloy was not included in these early sheathing trials because its composition with regard to iron and manganese was not yet established.
In the early trials, the and the 70-30 copper-nickel alloy sheaths were welded directly to the steel. One might assume that corrosion of the anodic steel below mean low tide would be accelerated because it is in direct contact with the much more noble sheathing material. A number of experiments were conducted at the LaQue Center to investigate this. Kirk has summarized these well (18) . It turns out, that in the tidal zone, steel is by far the most cathodic material, and the more noble sheathing alloys result in lower currents and corrosion of submerged steel than no sheathing at all.
This conclusion is illustrated nicely in the results of galvanic corrosion tests conducted to determine the effects on submerged steel coupled to other alloys in the tidal zone as shown in Figure 19. Plates of the alloys placed in the tidal zone are coupled to submerged steel plates, and the total current per tide was measured periodically over the 14-months of exposure. Current decreased with time, but the results demonstrated clearly that the most severe galvanic couple is steel to steel. This is because even though the potential difference developed in the noble alloy-to-steel couples is significantly greater than between two steel panels, the more rapid and more nearly complete polarization of the noble metals results in a great reduction in galvanic current. More recently, the International Copper Research Association (now the International Copper Association) conducted several research programs clarifying and elaborating on these earlier findings (22-24) . In summary, steel under water corrodes less when in contact with noble metals in the tidal zone than when coupled to another panel of steel in the tidal zone.
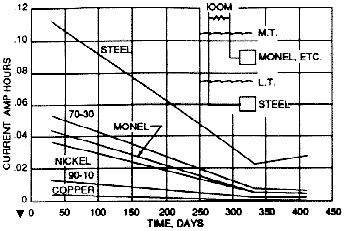 Figure 19 - Total current per tide vs. time between plates in a simulated piling test
Figure 19 - Total current per tide vs. time between plates in a simulated piling testFouling Protection
As discussed elsewhere in this series, the 90-10 copper-nickel alloy provides the best combination of corrosion resistance and biofouling protection. Attachment of this sheathing material to the steel structure by welding or mechanical fasteners will result in cathodic polarization of the sheath material and a reduction in the antifouling capability of the 90-10 copper-nickel alloy. Therefore it is necessary to electrically insulate the sheath from the steel jacket members to get full advantage of the properties of the alloy. Insulation can be achieved by pumping cement or an epoxy into the annular space between the component and the sheath or, more simply, by use of an elastomer or rubber-base insulator. The copper-nickel can be in the form of sheet, wire grid, particles (25) or flame-spray. Bonding of the steel-elastomer-copper-nickel interfaces can be by vulcanizing, by the use of epoxy adhesives, by mechanical means or a combination of methods. Estimated costs in 1986 ranged from $18 to $30 per square foot ($194 to $322 per square meter) (17).
Long term sheathed piling exposures to assess the effectiveness of corrosion and biofouling protection and measure the cathodic protection current required for several situations have been sponsored by the International Copper Association and the Copper Development Association, Inc. at the LaQue Center for Corrosion Technology, Wrightsville Beach, North Carolina (26). Over 50 ASTM Type A-36 piling 17cm in diameter were sheathed with 4.6 mm thick x 3 m long C70600. Some sheaths were directly welded to the steel, others were insulated from the steel with concrete or with 6 mm of a butyl rubber compound. Some piling were cathodically protected with Galvalum III anodes while others remained unprotected. Piling were removed after two years' and five years' exposure for study of corrosion of the steel and the fouling.
The results of biofouling accumulation on these pilings are summarized in Table 4, below. The mass accumulated on the bare steel piling was more than twice that on the directly welded C70600 and more than 50 times that attached to the insulated sheathing. Only a few scattered barnacles were seen on the unprotected copper-nickel sheaths after five years.
| Piling | kg/m² | Percent | |
|---|---|---|---|
| Bare Steel | 18.00 | 100.0 | |
| Concrete Insulated | 0.36 | 1.9 | Only scattered barnacle shells |
| Directly welded | 7.95 | 44.3 | |
| Rubber Insulated | 0.26 | 1.4 | Only scattered barnacle shells |
| Rubber insulated w/Galvanic Gouple (single point contact) | 4.59 | 25.5 | |
| Biofouling organisms observed - barnacles, oysters, codium, tunicate, colonial tunicate, encrusting and filamentious bryozoans | |||
The galvanic anodes used on the cathodically protected piling were cleaned and weighed; mass loss and consumption rates are given in Table 5, below. In the two year exposures, the directly welded piling displayed a lower anode consumption rate than the bare steel; the concrete insulated consumption rates was comparable to that of the bare steel. After five years of exposure, both the directly welded and the concrete insulated piling displayed reduced consumption rates. The reduction in anode consumption for the directly welded piling is considered to be due to the favorable polarization characteristics of the C70600 copper-nickel alloy. The reduced anode consumption rate for the concrete insulated piling is most likely due to the high resistance path through the concrete to the underlying steel.
| Piling | Weight Loss (g) | Consumption Rate (kg/yr) |
|---|---|---|
| TWO YEAR REMOVALS | ||
|
Bare Steel |
716.4 | 0.36 |
|
Concrete Insulated |
755.3 | 0.38 |
|
Directly Welded |
414.1 | 0.21 |
| FIVE YEAR REMOVALS | ||
|
Bare Steel |
1880.6 | 0.38 |
|
Concrete Insulated |
1256.6 | 0.25 |
|
Directly Welded |
687.6 | 0.14 |
| * Two Anodes Per Piling | ||
The overall reduction in anode consumption rates for both sheathing techniques could be due to the reduction in current required to maintain a calcareous film formed on the C70600 copper-nickel alloy.
It was also observed that even in the directly welded sheathing technique where piling were exposed without cathodic protection for five years, there was no grossly accelerated attack of the steel immediately above or below the sheath. The average corrosion rates in the steel adjacent to the sheathing below the mean low tide point did not exceed 0.25 mm/y, no higher than the rate in the freely corroding, unsheathed steel control pilings. Of course, exposure of any steel piling without cathodic protection is not recommended.
A proprietary system called "Bio-Shield" developed by the Shell Development Company, has met with considerable success on offshore installations off the coast of California(27). Biofouling can be quite severe along the Southern California coastline. Bioshield consists of 1-1/2 mm thick 90-10 copper-nickel and a high density elastomer with the trade name Splashtron made by the Mark Tool Company of Lafayette, Louisiana. After a laboratory test program, this system was applied to the design of the 214-m water depth Eureka platform with 60 well conductors (0.6 m OD). A total of 152 tonnes of structural steel, otherwise required to handle the loading from the marine growth, was eliminated. The platform was installed in July 1984. After several years, the copper-nickel surface of the Bioshield was free of fouling while the unprotected areas were covered with an 8 to 13 cm thick layer of barnacle and mussel growth. Platform responses were reduced: wave forces, -6%; base shear -10%; overturning moment, -10%; deck deflection, -10%; and pile load, -7%. The reduced platform response values are the reasons that prevention of marine growth can clearly reduce platform costs. Money was saved in reduced steel for corrosion allowance and improved fatigue characteristics in the major platform joints. Estimated savings realized from installing the sheathing system on the 60 conductors from +1.5 m mean low water line to -4.9 m for the 214 m structure were as follows in (1983-84):
| Conductors - 55 tons x $1000/ton | = | $ 55,000 |
|
Paint - 10,600 ft2 x $3/ft² |
= | $ 32,000 |
| Anodes - $1250 each x 4 | = | $ 5,000 |
| Structural Nodes - 114 tons x $2500/ton | = | $ 285,000 |
| Total | = | $ 377,000 |
Estimated savings of $50,000 to $100,000 per cleaning was also to be realized. Installed costs for this system on the Eureka platform were reported to be $250,000 or about $31.60/ft² ($340/m²). Clearly, this installation of a copper-nickel sheath system is this example was very cost effective.
Exxon Economic Analysis
The Exxon Production and Research Company carried out a generalized economic evaluation (26) for INCRA by means of a computer aided design study of a conventional steel structure as depicted in Figure 20. Only insulated copper-nickel alloy sheathing systems were considered as these gave the full economic benefits resulting from prevention of both marine fouling and corrosion. Design models were worked out for a range of situations, covering three different water depths, environmental conditions (wind, wave and current) ranging from mild to severe, and marine growth ranging from light to heavy. In all, 29 scenarios were considered. Potential cost savings were calculated based on the savings in weight of installed steel. The cost of the sheathing and its installation were not included in the analysis because of their variability with means of attachment.
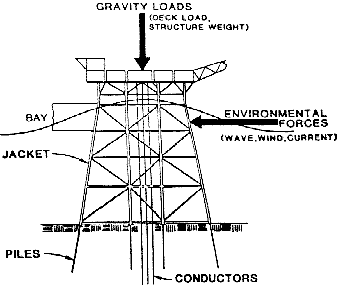 Figure 20 - Simplified diagram of forces acting on offshore structures
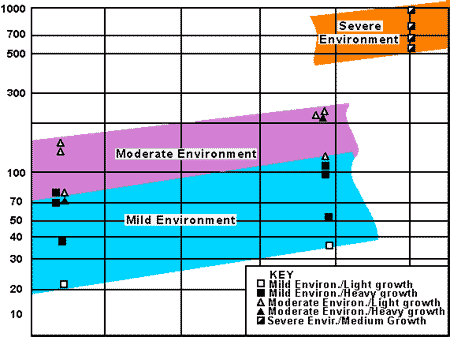
Figure 20 - Simplified diagram of forces acting on offshore structuresThe results of calculations of gross savings on the costs of materials, fabrication and installation for offshore structures deployed under each of the scenarios considered are summarized in Figure 21. In the mild environment, total weight savings ranged from 9 tonnes up to 174 tonnes for the various cases, with cost savings up to a maximum of 5% of the total structure cost. In the moderate environment, weight savings ranged from 80 to 404 tonnes and cost savings from 1.8% to 5.9% of total cost. The corresponding figures for the severe environment were 732 to 2372 tonnes weight saving and 2.9% to 9.3% cost savings.
Ratio of Potential Savings and Sheathing areas
($/M 2)
 Water Depth (M)
Water Depth (M)
Figure 21 - Potential Savings per Unit Area of Sheathing for Various Environme
British Gas Experience, Morecambe Bay, UK
The structures deployed in Phase One of the Morecambe Bay gas field project were sheathed with 90-10 copper-nickel alloy by welding 4 mm thick plate directly to the steel legs over the tidal and splash zones from 2 m below low tide level to 13 m above. A production platform, an accommodation platform, three drill platforms and a flare stack have been so treated. The main purpose of the sheathing was to provide corrosion protection in the splash zone. The submerged portion of the structure is protected by zinc anodes attached directly to the steel. An economic assessment(29) indicated that the 90-10 copper-nickel alloy sheathing was more cost effective than either the Monel (Trademark of the International Nickel Co.) 400 alloy sheathing or conventional systems using non-metallic coatings with increased steel thickness previously used on British Gas structures.
The certifying authorities required sacrificial steel (12 mm thickness) in this highly corrosive area when a paint system or neoprene wrap is specified. Sacrificial steel is not required with a copper-nickel (or Monel) metal wrap system. The economic justification was based on a platform life of 15 years. All maintenance costs were discounted to net present value at 10%. Costs were summarized as follows:
| Protective Coating/Sheathing | ||||
|---|---|---|---|---|
| Paint | Neoprone | Monel | 90-10 Cu-Ni | |
| Initial Cost - Extra Steel | 2.3 | 2.3 | - | - |
| Protective Material Labor | 0.1 | 0.3 | 2.2 | 0.95 |
| Maintenance Cost | 2.4(a) | unknown(b) | 0.15(c) | 0.15(c) |
| Extra Weight (tonnes) | 660 | 660 | 180 | 180 |
| (a) Repainting 8 years after installation and every 5 years thereafter (b) No long-time experience; no large scale repairs assumed in less than 18 years (c) Minimum maintenance, confined mainly to accident repair |
||||
After over two years in service it appears that the copper-nickel alloy sheathing on the Morecambe Bay structures is free of significant corrosion and, despite the fact that the sheathing was attached directly to the steel, there is less marine fouling than would have been expected if there had been no sheathing.
The application described shows a 90-10 copper-nickel sheathing system to have a net cost savings of from 52 to 77 percent compared to other systems evaluated.
Summary
Test programs on sheathing of offshore piling have shown the steel underlying the sheathing is virtually completely protected. Biofouling of insulated 90-10 copper-nickel sheathing greatly reduces the potential for structure loading due to increased weight and cross sectional area in turbulent waters. An appreciable decrease in the initial cathodic protection current required by the sheathed piling as compared to the bare steel piling has been observed.
Sheathing of support members and conductors of large offshore platforms has been commercially applied. A number of large installations have performed as expected with documented cost savings compared to the unsheathed design.
References
- S.W. Katsev, M.L. Katsev, "Last Harbor for the Oldest Ship," National Geographic, 146, No. 5, Nov. 1974 pp. 618-625.
- J.L. Manzolillo, E.W. Thiele and A.H. Tuthill, "CA-7-6 Copper-Nickel Alloy Hulls: The COPPER MARINER'S Experience and Economics." SNAME Annual Meeting, November 1966.
- "Corrosion Resistance of Wrought 90-10 Copper-Nickel-Iron Alloy Marine Environments," The International Nickel Co., Inc., Feb. 1975.
- M. Prager, J. Keay and E.W. Thiele, "Joining Copper and Copper-and Copper-Nickel-Clad Steels," Welding Journal, September 1978.
- E. Schorsch, R.T. Bicicchi and J.W. Fu, "Hull Experiments on 24-Knot R0/R0 Vessels Directed Toward Fuel-Saving Application of Copper-Nickel," SNAME Annual Meeting Paper, November 1978.
- "Copper-Nickel Sheathing Study - Phase II, Two Year Service Performance of Test Panels on the Arco Texas," U.S. Department of Transportation Maritime Administration, Report No. MA-RD, May 1984
- P. Drodten and H. Pircher, "Behavior of CuNi10FelMn-Clad Ship Plates in Sea Water," Werkstoffe und Korrosion41 (1990), pp 59-64.
- F.H. Todd, "Skin Friction Resistance and the Effects of Surface Roughness," Trans. Soc. of Naval Arch. & Marine Engineers, 59 (1951).
- H. Lackenby, "The Resistance of Ships, with Special Reference to Skin Friction and Hull Surface Condition," Institute of Mechanical Engineers, T.L. Cray Lecture, 1962.
- R.P. Nelson, E.G. Palumbo and R.D. Sedat, "Comparison of Three Methods of Coating Ship Surfaces to Control Underwater Roughness," Center for Maritime Studies, Webb Institute of Naval Architecture, November 1983.
- "Copper-Nickel Sheathing Cost Study - Phase III, Installation Methods and Cost Reduction Potential," U.S. Department of Transportation Maritime Administration, Report No. MA-RD-770-87026, August 1987.
- L.W. Sandor, "Pulsed MIG Spot Welding of Copper-Nickel to Steel for Ship's Hulls," Final Report, INCRA Project No. 352, INCRA, March 1983.
- Offshore, May 1990, p. 32.
- "Copper-Nickel Cladding for Offshore Structures," Publication TN37, Copper Development Association, Potters Bar, England, 1986.
- B.B. Moreton, "Copper-Nickel Sheathing of Offshore Jacket Structures," Australian Corrosion Association Conference 26, Adelaide, Australia, November 1986.
- R.W. Ross, Jr. and D.B. Anderson, "Protection of Steel Piling in Marine Splash and Spray Zones, - The Metallic Sheathing Concept," Proceeding of the Fourth International Congress on Marine Corrosion and Fouling, Antibes, France, June 1976, pp 461-473.
- C.J. Gaffoglio, "Concepts in Corrosion and Biofouling Control Using Copper-Nickel," Proceedings of the First OMAE specialty Symposium on Offshore and Arctic Frontiers, New Orleans, Louisiana, Feb. 23-27, 1986.
- W.W. Kirk, "Metallic Sheathing for Protection of Steel in Seawater," Materials Performance, 26 (No. 9), September 1987, pp 23-28.
- P.T. Gilbert, "Use of Copper-Nickel Alloy Sheathing for Corrosion and Fouling Protection of Marine Structures," Proceedings of The Institute of Metals Conference on Marine Engineering with Copper-Nickel, London, April 1988, pp 21-41.
- E.V. Creamer, "Splash Zone Protection of Marine Structures," Offshore Technology Conference, Paper No. 1274, Houston, Texas, 1970.
- B.B. Morton, "Report on the Protection of Offshore Steel Structures by a Metallurgical Method, "9th Annual Conference Petroleum Division ASME," New York, N.Y., September 1954.
- Danish Corrosion Centre. INCRA Project No. 371. Report in preparation.
- J.A. Ellor & G.A. Gehring, Jr., "Galvanic Corrosion of Copper-Nickel Sheathed Steel Piling," Ocean City Research Corp., INCRA Project No. 368. March 1987.
- S. Husa, "Stray Current corrosion", Marintec, INCRA Project No. 372, September 1986.
- J.W. Brockbank, "Avonclad - A Flexible Neoprene/Copper-Nickel Material for Long Term Anti-Fouling: A Review of Manufacture, Tests and Recent Installations," Proceedings of the Institute of Metals Conference on Marine Engineering with Copper-Nickel, London, April 1988, pp 101-103.
- D.G. Melton, "Review of Five-Year Exposure Data for CuNi-Sheathed Steel Pilings," Proceedings of the 23rd Annual Offshore Technology Conference, Houston, Texas, May 1991, pp 221-2 33.
- Engel & J.P. Ray, "Bio-Shield: An Anti-fouling System for Offshore Platforms that Works!," Proceedings of Ocean Engineering and the Environment Conference, San Diego, California, United States, Vol 1, 1985, p. 62.
- Barger, L.D. Downer, J.E. Brown and T.R. Gaul, "Economic Evaluation - Use of Copper-Nickel Alloy for Sheathing of Offshore Structures," Exxon Production Research Company, INCRA Project No. 359, September 1984, pp 21-41
- R. Carruthers, "The Use of 90/10 Copper-Nickel Alloy as a Splash Zone Cladding," Paper 6, Proceedings of CDA Conference on Copper-Alloys in Marine Environments, Birmingham, England, April 1985.