The Fate of Copper Released from the Vasa Ship Museum
Copper Applications in Health & Environment
"This document is reprinted, with permission from Metall, 52. Jahrgang, Nr. 4/98 p. 230-231"
Scandinavian Copper Development Association
Corrosion of Copper in Stockholm | Fate of Copper Around the Vasa Museum | Conclusions | References
Stockholm is a city close to lake and sea. It is also a city with many copper roofs, especially in the old town. Most of these roofs have the nice green-blue patina colour consisting of different copper minerals. The tradition of using copper sheet for roofs is at least 400 years old and is closely related to the copper production in Sweden at Stora Kopparberget. For example, the old royal palace in Stockholm, Tre Kronor, (Three Crowns) got a copper roof in the late 16th century. King Johan III thought the old wood and straw roofs were a fire hazard. When Stockholm grew and became a more continental city during the 17th century, the great power age of Sweden, a lot of new palaces and other stone houses were built and fitted with copper roofs. The Town Museum of Stockholm has a model showing the buildings towards the end of the 17th century. A lot of the houses have roofs of copper. The copper was sometimes subsidized by the government in order to increase the usage.
 The Vasa Museum
The Vasa MuseumCopper as material for roofs and other decorative parts on important building is still well estalblished in Stockholm. It was therefore natural that the new museum for the 17th century war ship Vasa got a roof of copper in 1990. The total area of roof and sidings is 12.000 m2. It has been suggested that large amounts of copper are released from the roof into the sea due to corrosion.
Corrosion of Copper in Stockholm 
The corrosion process of the copper surface, i.e. interaction between atmosphere and metal, and with time forms the familiar green-blue patina.
The corrosion rate has decreased by a factor of 3 during time period 1965 to 1994, see Figure 1. This is a clear indication of the improved air quality and the protection afforded by the formation of the patina. Figure 1 shows the total corrosion (mass loss consisting of adhearing and released corrosion products). The released amount, run off, during 1988-1995 has been estimated and for the studies beginning 1995 it has been measured. As can be seen there is very little variation in spite of the high total corrosion rate. The corrosion products that are released from the Vasa museum roof can be estimated to be 0.15 mm/year, or 1.3 g/year, or 0.025% of the sheet metal thickness. The maximum amount of copper released is thus 15.6 kg/year. The copper run off is in the form of partly desolved (ions) and partly copper bound to particles.
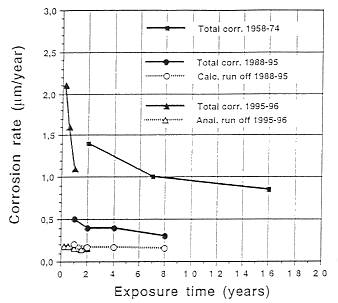 Figure 1. Corrosion of copper sheet in central Stockholm.
Figure 1. Corrosion of copper sheet in central Stockholm.Total corrosion, mass loss 1958-74 (1)
Total corrosion, mass loss 1988-95 (2)
Calculated run off 1988-95 (2)
Total corrosion, mass loss 1995-96 (3)
Fate of Copper Around the Vasa Museum 
The roof of the museum has no gutters and down pipes. Concrete mixed with stones has been laid under the eaves around the building in order to prevent the rain water from destroying the lawns.
It was observed the spring 1996 that the concrete had green-blue patches and pieces of the concrete (Figure 2) were gathered for closer study. X-ray diffraction and scanning electron microscopy (SEM-EDS) were used on surface pieces with strong and weak colour. Strong color resulted from the formation of the two copper minerals malachite and azurite (by analysis, 30% copper, 13% silicon, 14% calcium, 0.7% sulphur. The oxygen content could not be determined). The pieces with weaker color had the compositon 0.5% copper, 24% silicon, 17% calcium, 0.3% sulphur. The concrete pieces were green-blue through their whole thickness.
 Figure 2. Pieces of concrete from the string around the museum.
Figure 2. Pieces of concrete from the string around the museum.The amount of copper that has been bound to the concrete can be estimated as follows: The density of the concrete is 2.3 kg/dm3 therefore, the total copper present is 11.5 g/dm3, for the part with the copper content 0.5%. The surface layers are estimated to 36 dm2/dm3 concrete with a thickness of 0.02 mm. The copper amount in the surface layer is therefore 8.4 g/dm3 and the total amount is 10-20 g/dm3. 1 dm string is around 1 dm3 concrete and the total length is 300 m i.e. the copper amount 30-60 kg.
 Figure 3. Marks from run off rain water on plastered wall with copper cover.
Figure 3. Marks from run off rain water on plastered wall with copper cover.The reaction between copper ions in rain water and concrete, especially with the calcium carbonate (CaCO3), is very fast. Figure 3 shows a plastered wall with a cover of copper on the top. Rain water flowing down the plaster surface has carried copper with it to create the green-blue discolourations which stops half way down the wall. All the dissolved copper has reacted with CaCO3. The calcium has exchanged with copper and the blue-green malachite and azurite minerals have formed.
Conclusions 
The roof of the Vasa ship museum has hardly given any bioactive copper (ions) to the ground or, more importantly, the sea, around the building during its first 6 years. All ions leaving the roof in rain water run-off have reacted with the concrete on the ground and been bound in mineral form. The rest of the copper in the run off is in particle form and not as reactive and also not bioavailable to terrestial or aquatic organisms. The particles are even more quickly and strongly bound when in contact with soil or organic matter.
The green-blue coloured concrete or stone that can be seen on pavements where rain water from copper roofs and down pipes is running out is easily explained. It is the copper ions that react with calcium containing material. The green-blue colour stops in many cases half way across the pavement even though the rain water has flowed beyond the concrete area. This is an indication that all copper ions have reacted. Even in this case has no bioactive ions reached the street or ground.
References:
1. Holm, R., Mattson, E. Atmospheric Corrosion Tests of Copper and Copper Alloys in Sweden. 16 Years Results ASTM Sp. Tech. Publ. 767, 1982.
2. Persson, O., Kucera, V. Metallutsläpp orsakade av korrosion och nedbrytning av olika materialytor. Swedish Corrosion Institute KI 1996:3
3. Odnevall, I, Leygraf, C. Project still in progress at Corrosion Department, Royal Inst of Technology, Stockholm, Sweden.
Also in this Issue:
- The Fate of Copper Released from the Vasa Ship Museum
- The Swedish Program for Long Term Isolation of High Level Nuclear Waste in Copper Canisters
- The Copper Motor Rotor: New Technology for High Efficiency Motors
- Shape Memory and Superelastic Alloys
- Copper's Role in Radioactive Waste Disposal III - The US Experience
